 Neutron Therapeutics, a targeted radiation therapy company developing a comprehensive solution for Boron Neutron Capture Therapy (BNCT), and Cosylab, the world’s leading provider of control systems for the planet’s most complex machines, today announced that Neutron Therapeutics’ nuBeam BNCT System, using Cosylab’s OncologyOne software, has reached a significant milestone in its clinical commissioning at Helsinki University Hospital – first simulated patient treatment.
Neutron Therapeutics, a targeted radiation therapy company developing a comprehensive solution for Boron Neutron Capture Therapy (BNCT), and Cosylab, the world’s leading provider of control systems for the planet’s most complex machines, today announced that Neutron Therapeutics’ nuBeam BNCT System, using Cosylab’s OncologyOne software, has reached a significant milestone in its clinical commissioning at Helsinki University Hospital – first simulated patient treatment.
BNCT is a targeted radiation cancer therapy in which neutron beams destroy only boron compound-bearing tumors without destroying neighboring normal tissue. BNCT has the potential to deliver highly effective and cell-localized radiation therapy to treat tumors with minimal impact on the patient’s quality of life compared to other radiation, chemotherapy or biological treatment modalities currently in use.
Neutron Therapeutics Inc. and the Helsinki University Hospital are collaborating to launch the first European hospital-based BNCT facility. Cosylab is a proven partner with over two decades of experience solving complex software challenges in radiation therapy.
Johanna Mattson, Senior Medical Director at the Helsinki University Hospital Comprehensive Cancer Center, commented: “Providing BNCT with the most sophisticated accelerator-based device will enable Finnish clinicians to treat patients with some of the most obstinate cancers and remain globally at the forefront of oncology. We are working hand in hand with our industry partners, Neutron Therapeutics and Cosylab, to bring the full clinical potential of BNCT to patients in Helsinki as soon as 2023.”
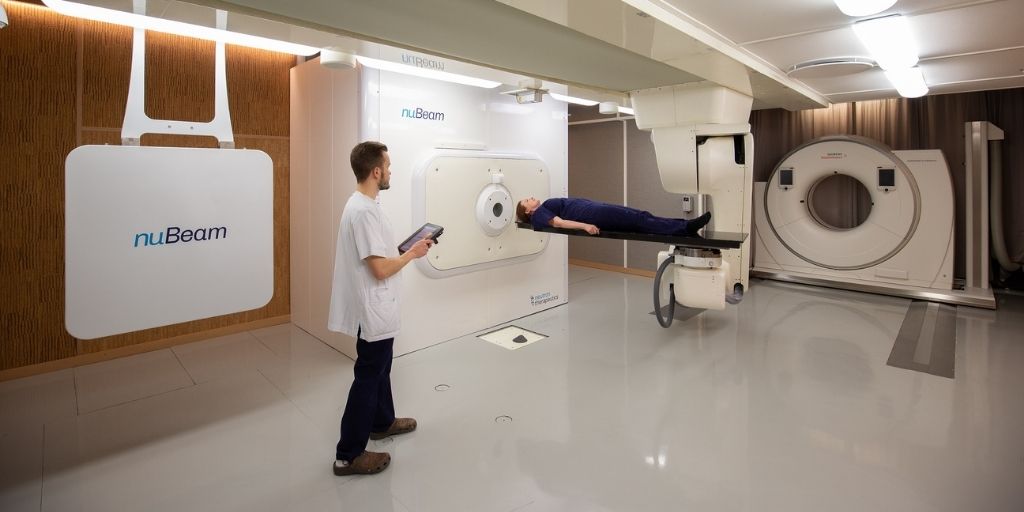
Neutron Therapeutics nuBeam is a high-throughput, compact accelerator-based neutron source suitable for clinical settings. nuBeam replaces legacy nuclear reactors. It has the highest neutron flux of all currently available BNCT systems and is the only device producing an IAEA-compliant BNCT beam for the safe and effective clinical use of neutrons. During the commissioning process, the Helsinki nuBeam device has demonstrated both robustness and reliable operation, validating the Neutron Therapeutics technology as the best choice for a high-throughput BNCT clinic with stringent safety requirements.
“We are extremely excited that our collaboration with Helsinki University Hospital has demonstrated nuBeam’s excellent reliability in the clinical environment. We are committed to achieving the initiation of clinical trials in the first half of 2023 at our first European nuBeam installation. Our close partnership with Cosylab has enabled rapid progress towards this goal and positions Neutron Therapeutics to maximize the potential of BNCT and its beneficial impact on patients,” said Dr. Elizabeth Reczek, CEO of Neutron Therapeutics.
The first simulated patient treatment at the Helsinki nuBeam installation was achieved using Cosylab’s OncologyOne software, the only solution on the market that covers all the needs of a radiation therapy device. It has already proven itself in other forefront radiation therapy modalities such as proton therapy and brings to the nuBeam system a software solution for radiation therapy that is much more readily upgradable and integrable than custom-built software while guaranteeing the shortest time-to-clinical-use.
“We are thrilled by the first-beam success and proud we could help Neutron Therapeutics stay on schedule with the commissioning of their nuBeam in Helsinki. Our OncologyOne suite of software products is designed from the ground up to empower our customers to bring their radiation therapy innovations into clinical practice in the shortest possible time and full working order, stated Dr. Mark Pleško, CEO at Cosylab.
Finland has been a European hotspot of BNCT trials since 1992. With the ongoing clinical verification and validation testing of the nuBeam system at the Helsinki University Hospital, patients who have inoperable, locally recurrent head, neck, and other cancers incurable with conventional radiation therapy are now much closer to reaping benefits from boron neutron capture therapy.